 ōFolding of
the Villin Headpiece Subdomain from Random Structures. Analysis of the Charge
Distribution as a Function of the pH.ö D.R. Ripoll, J.A. Vila and H.A.
Scheraga. (2004). J. Mol. Biol. 339, 915-925.
[pdf]
ōFolding of
the Villin Headpiece Subdomain from Random Structures. Analysis of the Charge
Distribution as a Function of the pH.ö D.R. Ripoll, J.A. Vila and H.A.
Scheraga. (2004). J. Mol. Biol. 339, 915-925.
[pdf]This project is carried in collaboration with H. A. Scheraga and members of his group at the Department of Chemistry at Cornell.
 ōFolding of
the Villin Headpiece Subdomain from Random Structures. Analysis of the Charge
Distribution as a Function of the pH.ö D.R. Ripoll, J.A. Vila and H.A.
Scheraga. (2004). J. Mol. Biol. 339, 915-925.
[pdf]
ōFolding of
the Villin Headpiece Subdomain from Random Structures. Analysis of the Charge
Distribution as a Function of the pH.ö D.R. Ripoll, J.A. Vila and H.A.
Scheraga. (2004). J. Mol. Biol. 339, 915-925.
[pdf]
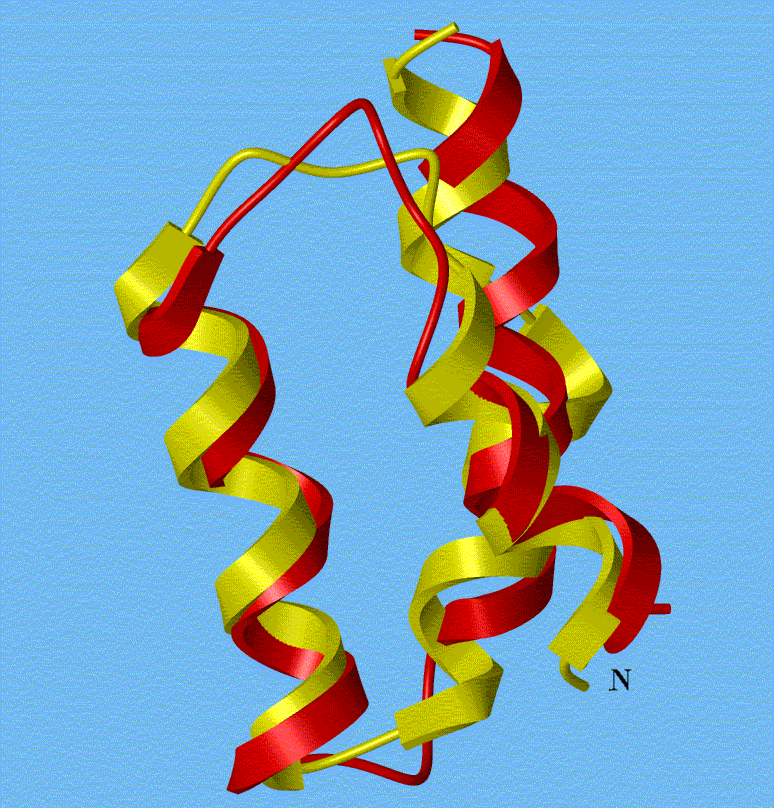 ōAtomically
detailed folding simulation of the B domain of staphylococcal protein A from
random structures.ö
J.A. Vila, D.R. Ripoll, and H.A. Scheraga. (2003). Proc. Natl
Acad. Sci. USA. 100, 14812-14816.[pdf]
ōAtomically
detailed folding simulation of the B domain of staphylococcal protein A from
random structures.ö
J.A. Vila, D.R. Ripoll, and H.A. Scheraga. (2003). Proc. Natl
Acad. Sci. USA. 100, 14812-14816.[pdf]
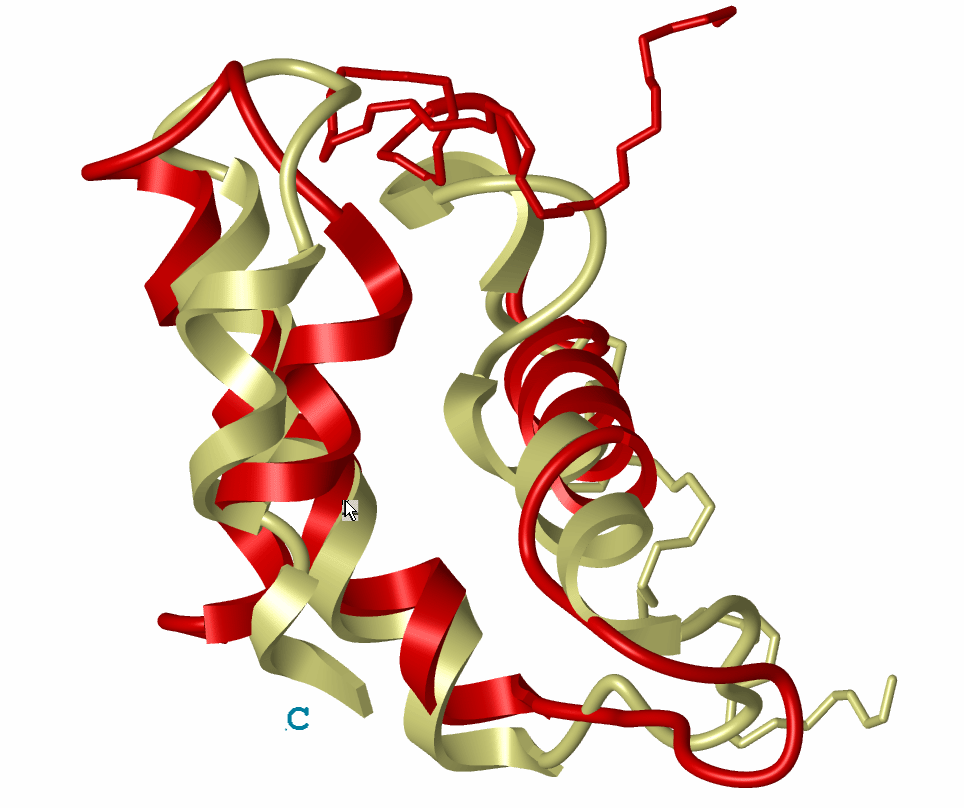 A. Liwo, J. Lee, D.R. Ripoll, J. Pillardy, and H.A. Scheraga. (1999). ōProtein Structure
Prediction by Global Optimization of a Potential Energy Functionö.
Proc. Natl. Acad. Sci. USA, 96, 5482-5485.
[pdf]
A. Liwo, J. Lee, D.R. Ripoll, J. Pillardy, and H.A. Scheraga. (1999). ōProtein Structure
Prediction by Global Optimization of a Potential Energy Functionö.
Proc. Natl. Acad. Sci. USA, 96, 5482-5485.
[pdf]
J. Lee, A. Liwo, D.R. Ripoll, J.Pillardy, J.A. Saunders, K.D. Gibson, and H.A. Scheraga.. (2000). ōHierarchical energy-based approach to protein-structure prediction; blind-test evaluation with CASP3 targetsö. Int. J. Quant. Chem. 77, 90-117. [pdf]
``A Molecular Switch for Biological Logic Gates: Conformational Studies.'' G. Ashkenazi, D. R. Ripoll, N. Lotan, H. A. Scheraga. (1997). Biosensors & Bioelectronics 12, 85-95. [pdf]
 J.A. Vila, D.R. Ripoll and H.A. Scheraga. (2001). ōInfluence of Lysine
Content and pH on the Stability of Alanine-Based Co-polypeptidesö.
Biopolymers, 58, 235-246.
[pdf]
J.A. Vila, D.R. Ripoll and H.A. Scheraga. (2001). ōInfluence of Lysine
Content and pH on the Stability of Alanine-Based Co-polypeptidesö.
Biopolymers, 58, 235-246.
[pdf]
J.A. Vila, D.R. Ripoll and H. A. Scheraga. (2000). ōPhysical reasons for the unusual α-helix stabilization afforded by charged or neutral polar residues in alanine-rich peptidesö. Proc. Natl. Acad. Sci. USA. 97, 13075-13079. [pdf]
Coupling between Folding and Ionization Equilibrium. Effects of pH on the Conformational Preference of Oligopeptides. D. Ripoll, Y. Vorabjev, A. Liwo, H. Scheraga and J. Vila. (1996). J. Mol. Biol. 264, 770-783. [pdf]
To see another figure showing the electrostatic field and surface potential generated by one of the lowest energy conformation click here.
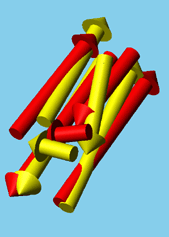
M. Nanias, M. Chinchio, J. Pillardy, D.R. Ripoll, and H. A. Scheraga. (2003). ōPacking helices in proteins by global optimization of a potential energy function.ö Proc. Natl. Acad. Sci. USA, 100, 1706-1710. [pdf]